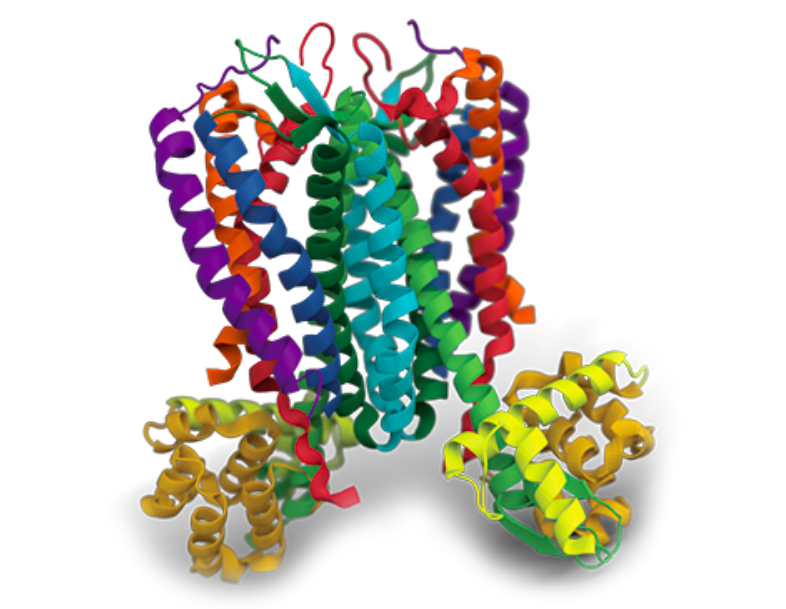
Cell-free protein synthesis (CFPS) methodology offers the possibility to produce proteins that would be difficult or even impossible to synthesize in cell-based systems. These so-called “difficult-to-express proteins” include membrane proteins, but also cytotoxic proteins (toxic cancer therapeutics, antimicrobial peptides/proteins and toxins), vaccines, antibodies and proteins containing non-standard amino acids [1]. Cell-free systems are, also, advantageous for the synthesis of proteins with biased coding sequences or with defined post-translational modifications, for the ones that require chaperones or specific chemical environments and even for the synthesis of virus-like particles and bacteriophages.
Bruno Tillier

Recent Posts
Mythbusting#1: Cell-free systems are only relevant for membrane proteins
Topics: Cell-free technology, Proteins
Proteoliposomes - ideal model systems for membrane protein analysis
Proteoliposomes – ideal model systems to study the structure and function of membrane proteins in semi-native environment by cryogenic electron microscopy
Topics: Proteins, Proteoliposomes
Five reasons to label your proteins for NMR studies using cell-free systems.
Isotope labelling has been used for over 40 years to facilitate the study of protein structure using nuclear magnetic resonance (NMR) spectroscopy. During that time, traditional expression systems such as in vivo protein synthesis were the preferred options for fabricating the labelled proteins. However, nearly a decade ago, it was discovered that cell-free protein expression offers an abundance of beneficial properties making it more adapted to the addition of isotopes than first thought. Here, we give you our top five reasons to use a cell-free system to label your proteins for your NMR study…
Topics: Cell-free technology, Proteins
Mythbusting #9 : “Cell-free systems do not produce functional protein complexes"
The open nature of cell-free systems allows the production of protein complexes much more easily than with in vivo systems.
Topics: Cell-free technology